Recommended videos
Recommended videos
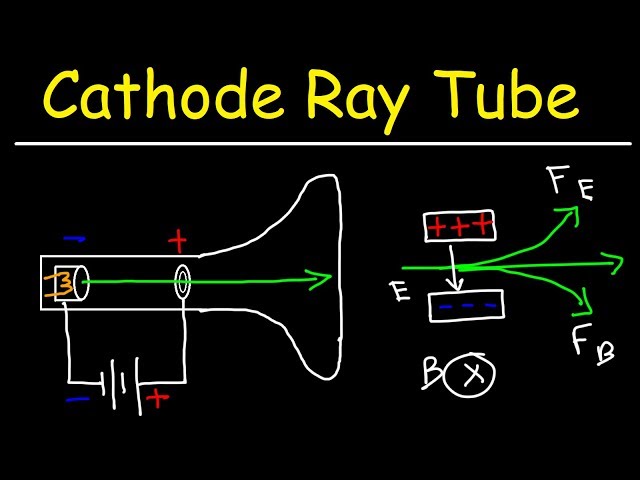
Cathode Ray Tube Experiment and Charge To Mass Ratio of an Electron
164,601 views
4 years ago

The Cathode ray oscilloscope C.R.O-Physics-Cathode rays
105,033 views
5 years ago

164,601 views
4 years ago

105,033 views
5 years ago
117 Comments